We have much more to do and your continued support is needed now more than ever.
Predator Impacts on the Spread of Chronic Wasting Disease
Chronic wasting disease (CWD) is a fatal prion disease — a disease caused by misfolded proteins in the brain — impacting cervids, such as deer, elk, and moose, across the U.S. with occurrences documented in 30 states since its discovery in the 1960’s (USGS).
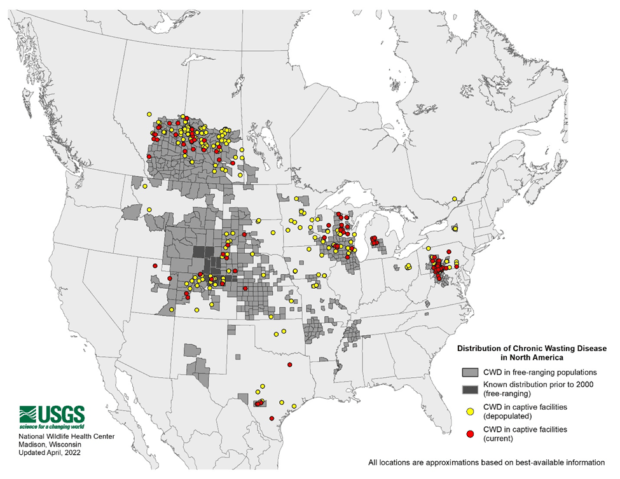
As CWD has increased in prevalence, so has its attention from wildlife conservation entities, state and federal governments, and the general public for finding solutions to curb its spread. Possibilities regarding the impacts of cervid predation by large carnivores, namely wolves and mountain lions, on the spread of CWD have become of particular interest, especially in the media and other realms of public discourse. In order to make the most informed decisions in our communications and advocacy for CWD research and management, National Wildlife Federation has conducted a review of current literature and expertise to most accurately inform our understanding of what is currently known about the impact of predation on the disease ecology of CWD.
The messaging in media and public discourse has largely overstated the idea that large predators, especially wolves, are the solution for curbing the spread of CWD, ignoring the uncertainty in the literature. The current literature on the relationship between cervid predation and the spread of CWD lacks the empirical data needed to make strong predictions outside of local contexts.
There are several challenges and considerations in making predictions about the impacts of predation on the prevalence and spread of CWD. First, systems hosting infected populations of cervids differ widely–dynamics that are true in one system cannot necessarily be used to make accurate predictions in another system. These differences may include differences in prey species (such as white-tailed deer, mule deer, and elk), host contact rates, and the amount of time that a reservoir of CWD prions has existed in a given environment.
Second, in prey species, vulnerability to disease must overlap with vulnerability to predation; predators must select for infected individuals when they hunt. This overlap is necessarily dependent on variables such as the predator species, the prey age preference of predators, and differential vulnerability to CWD between age classes of the prey.
Predators would have the greatest impact on decreasing the prevalence of CWD in a given ecosystem if the most infectious individuals in a prey population are also the most vulnerable to predation (Brandell et al. 2022). Additionally, predators and prey need to overlap spatially — with gray wolves, for example, this reality is absent in many North American ecosystems where CWD is present.

Can predators reduce transmission?
Several studies have assessed the relationships between predation and CWD prevalence in local contexts and with specific predator species. For example, in the northern Front Range of Colorado, mountain lions selectively preyed upon infected mule deer — a dynamic that we might expect to decrease CWD prevalence (Krumm et al. 2010). However, in the ecological context and conditions of Table Mesa, Colorado, this selective predation of infected prey by mountain lions did not curb the transmission of CWD, possibly due to the increased congregation of prey to avoid predators, or the potentially cyclical nature of the epidemic (Miller et al. 2008). This dynamic may have also been due to the fact that the level of selective predation was simply not high enough to reduce transmission.
In a model developed using empirical data from the Greater Yellowstone Ecosystem, gray wolves and mountain lions may decrease CWD outbreak size substantially and delay the accumulation of symptomatic deer and elk. However, this effect is driven by their ability to select prey in the late-stages of CWD disease progression, which may or may not be realized on the landscape (Brandell et al. 2022).
In addition to the impacts of predators’ prey selection, the life history differences between prey species can differ substantially, as is seen in the differences between white-tailed deer, elk, and mule deer (the latter two species being the focus of most literature on the subject). It is also important to note that predator numbers in a system like the Greater Yellowstone Ecosystem are higher than social tolerance would allow in most other regions.
In addition to the potential for predators to decrease the spread of CWD, there also exists the possibility that predators and scavengers may contribute to the geographic spread of CWD–a spread that has not been entirely contiguous. In experimental conditions, mountain lions passed ~3% of the CWD prions they consumed–a pattern that may contribute to the spread of prions in the environment, but ultimately eliminating/sequestering most of the prions they consume (Baune et al. 2021).
In experimental conditions with mouse-adapted scrapie, American crows, a common scavenger across North America, have also passed infectious prions in their feces after being fed infected material, indicating that they may also translocate infectious prions that cause CWD (VerCauteren et al. 2012). Scavengers like crows may have the greatest impact when roosting in large numbers, defecating, and dropping pellets in a concentrated area. However, they may also have a diluting effect on the accumulation of CWD locally by dispersing the prions from the location where the infected animal died, lessening the reservoir (Fischer et al. 2013).
Hunter harvests
Parallel to the consideration of large predator impacts on the spread of CWD are the potential effects of human hunters. In Western states, hunters have had measurable impacts on the spread of CWD. Harvests that fell closer to breeding seasons were associated with lower CWD prevalence in male mule deer the next year (Conner et al. 2021).
Across 12 areas in Colorado, those with the greatest decrease in annual hunting license numbers (predation pressure) showed the greatest increases in the proportion of infected adult male deer killed by hunters (Miller et al. 2020). When sufficient in magnitude and sustained, hunter harvests may be effective in decreasing CWD prevalence in adult male mule deer, especially in the early stages of an epidemic (Conner et al. 2021).
Just as is the case with large predators, ecological context matters, along with social context. Hunting regulations, hunting practices (which may not change with hunting regulations), and access to public hunting lands may result in geographic differences in the impact of hunters on CWD prevalence.

Making informed management decisions
To address the uncertainty in models that predict the impacts of predators on CWD spread — and subsequently make informed management decisions — there are several needs and considerations. First, collecting data about the movement and predation patterns of large predators requires time and resources (in finances and personnel) — substantially more than is currently available to experts studying the disease ecology of CWD. This empirical data is critical for ground-truthing predictive models.
Second, the impact of scavengers on the spread of CWD — while likely having a relatively small effect compared to the movement of infected animals or human-mediated movement of infected carcasses — is not well understood, and would benefit from research investments to help conservationists have a more robust understanding of how CWD may spread geographically.
Finally, public opinion is a social variable that impacts both research and management decisions, both in the management of predators and the management of prey. For example, localized culling of wild cervids has been shown to decrease CWD spread soon after outbreaks are identified but tends to be widely opposed by the public (Joly et al. 2006; Manjerovic et al. 2014)
Leading, trusted conservation organizations with large constituencies, like the National Wildlife Federation, can be important messengers and advocates in the effort to accurately respond to and communicate information about CWD, predators, and the resources needed to better understand their relationship.
Considering the literature on the relationship between chronic wasting disease and predation of cervids by large predators, it is our responsibility to contribute to a balanced public discourse that communicates the lack of certainty in our general understanding of this relationship, the urgent need for increased capacity for data collection and research, and illustrate a fair, non-polarizing predator narrative — one that does not make them either the “heroes” or the “villains.”
Join the National Wildlife Federation in our support of the Chronic Wasting Disease Research & Management Act (S. 4111) by urging your senators to pass this critical legislation as soon as they reconvene in November.
Works Cited
Baune, C., Wolfe, L. L., Schott, K. C., Griffin, K. A., Hughson, A. G., Miller, M. W., & Race, B. (2021). Reduction of Chronic Wasting Disease Prion Seeding Activity following Digestion by Mountain Lions. MSphere, 6(6). https://doi.org/10.1128/msphere.00812-21
Brandell, E. E., Cross, P. C., Smith, D. W., Rogers, W., Galloway, N. L., MacNulty, D., … & Hudson, P. J. (2022). Examination of the interaction between age‐specific predation and chronic disease in the Greater Yellowstone Ecosystem. Journal of Animal Ecology. https://doi.org/10.1111/1365-2656.13661
Conner M.M., Wood M.E., Hubbs A., Binfet J., Holland A.A., Meduna L.R., Roug A., Runge J.P., Nordeen T.D., Pybus M.J., Miller M.W. (2021). The relationship between harvest management and chronic wasting disease prevalence trends in western mule deer (Odocoileus hemionus) herds. Journal of Wildlife Diseases, 57(4), 831-843. 10.7589/JWD-D-20-00226
Fischer, J. W., Phillips, G. E., Nichols, T. A., VerCauteren, K. C. (2013). Could avian scavengers translocate infectious prions to disease-free areas initiating new foci of chronic wasting disease?. Prion, 7(4), 263-266. https://doi.org/10.4161/pri.25621
Joly, D.O.,Samuel, M.D., Langenberg, J.A., Blanchong, J.A., Batha, C.A., Rolley, R.E., Keane, D.P. , Ribic, C.A. (2006). Spatial epidemiology of chronic wasting disease in Wisconsin white-tailed deer. Journal of Wildlife Diseases, (3),578–588. https://doi.org/10.7589/0090-3558-42.3.578
Krumm, C. E., Conner, M. M., Hobbs, N. T., Hunter, D. O., Miller, M. W. (2010). Mountain lions prey selectively on prion-infected mule deer. Biology Letters, 6(2), 209–211. https://doi.org/10.1098/rsbl.2009.0742
Manjerovic, M. B., Green, M. L., Mateus-Pinilla, N., & Novakofski, J. (2014). The importance of localized culling in stabilizing chronic wasting disease prevalence in white-tailed deer populations. Preventive Veterinary Medicine, 113(1), 139-145. https://doi.org/10.1016/j.prevetmed.2013.09.011
Miller, M. W., Runge, J. P., Holland, A. A., & Eckert, M. D. (2020). Hunting pressure modulates prion infection risk in mule deer herds. Journal of Wildlife Diseases, 56(4), 781-790. https://doi.org/10.7589/JWD-D-20-00054
Miller, M. W., Swanson, H. M., Wolfe, L. L., Quartarone, F. G., Huwer, S. L., Southwick, C. H., & Lukacs, P. M. (2008). Lions and prions and deer demise. PLoS ONE, 3(12). https://doi.org/10.1371/journal.pone.0004019
National Wildlife Health Center (2022). Expanding Distribution of Chronic Wast ing Disease. USGS. ttps://www.usgs.gov/centers/nwhc/science/expanding-distribution-chronic-wasting-disease
VerCauteren, K. C., Pilon, J. L., Nash, P. B., Phillips, G. E., & Fischer, J. W. (2012). Prion Remains Infectious after Passage Through Digestive System of American Crows (Corvus brachyrhynchos). PLoS ONE, 7(10). https://doi.org/10.1371/journal.pone.0045774





















